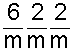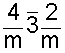< Return to Miles Mineral Museum Information
Graphite and diamond are two of the most interesting minerals. They are identical chemically – both are composed of carbon (C), but physically, they are very different. Minerals which have the same chemistry but different crystal structures are called polymorphs.
When you look at graphite and diamond, it is hard to imagine that they are identical chemically, for they are so different physically. Graphite is opaque and metallic- to earthy-looking, while diamonds are transparent and brilliant.
Another important physical difference is their hardness. The hardness of minerals is compared using the Mohs Hardness Scale, a relative scale numbered 1 (softest) to 10 (hardest). Graphite is very soft and has a hardness of 1 to 2 on this scale. Diamonds are the hardest known natural substance and have a hardness of 10. No other naturally occurring substance has a hardness of 10. The crystal structure of graphite yields physical properties that permit the use of graphite as a lubricant and as pencil lead. The gem and industrial properties of diamond, physical properties that we cherish and exploit, are also a result of diamond's crystal structure.
The reason for the differences in hardness and other physical properties can be explained with the molecular models below. In graphite, the individual carbon atoms link up to form sheets of carbon atoms. Each sheet of carbon atoms is translated (offset) by one-half of a unit such that alternate sheets are in the same position. Within each sheet every carbon atom is bonded to three adjacent carbon atoms that lie at the apices of equilateral triangles. This produces hexagonal rings of carbon atoms. Each carbon atom has four valence electrons available to participate in the formation of chemical bonds. Three of these electrons are used in forming strong covalent bonds with the adjacent atoms in the sheet. Covalent bonds are a type of chemical bond in which electrons are shared between atoms. The fourth electron is free to wander over the surface of the sheet making graphite an electrical conductor. The spacing between the sheets of carbon atoms is greater than the diameter of the individual atoms. Weak bonding forces called van der Waals forces hold the sheets together. Because these forces are weak, the sheets can easily slide past each other. The sliding of these sheets gives graphite its softness for writing and its lubricating properties.
In diamonds, each carbon atom is strongly bonded to four adjacent carbon atoms located at the apices of a tetrahedron (a three-sided pyramid). The four valence electrons of each carbon atom participate in the formation of very strong covalent bonds. These bonds have the same strength in all directions. This gives diamonds their great hardness. Since there are no free electrons to wander through the structure, diamonds are excellent insulators. The brilliance and "fire" of cut diamonds is due to a very high index of refraction (2.42) and the strong dispersion of light; properties which are related to the structure of diamonds.